High-Quality Targeted Alpha PSMA Therapy Cost In India
- The estimated Cost of Targeted Alpha PSMA Therapy In India ranges between USD 7000 into USD 12500.
- Based on the Stage of cancer, the achievement rate of Targeted Alpha Therapy is 80- 85 percent.
- The essential stay in the hospital of 5 days and 2 weeks beyond the hospital.
- Tests needed before the Therapy comprises Blood Tests, Pet CT Scan, and Biopsy.

Brief Overview
Alpha PRRT Therapy is based on the use of specific peptides and monoclonal antibodies, labelled with alpha-emitting radionuclides like Actinium (Ac225) or Bismuth (Bi213) or Lead (Pb212). The conjugated molecules target the cancer cells through the specific receptors which are located on the surface of the cancer cells and can selectively bind to these receptors. Once attached, these alpha-emitting radiomolecules destroy the cells by delivering radiation which kills the cells. Alpha particles have a high energy in the range of 5-9 MeV and at the same time a very short path length in human tissue below 0.1 mm, corresponding to less than 10 cell diameters. Consequently, the use of alpha emitters allows the specific targeting and killing of individual malignant cells, while minimizing the toxicity to surrounding healthy tissue.
How is Alpha PRRT Better than Conventional PRRT?
Targeted alpha PRRT has the potential to overcome many of the limitations of conventional Lu177 PRRT. As the alpha particles are more energetic than the B particles used in conventional PRRT, the alpha particles induce damage such as double-stranded DNA breaks and base chemical modifications. Unlike the damage caused by the beta particles, the kind of damage caused by the alpha particles is difficult to repair by the cancer cell, leading to more rapid cell death. Targeted alpha therapy thus has the ability to overcome the radioresistance to beta particles therapy. Also since the range of the alpha emitters is in millimeters in tissue, the surrounding normal tissue is unaffected by alpha emitters. Targeted alpha therapy often called the magic bullet therapy thus is an excellent alternative to treat patients of Neuroendocrine Tumours, especially those who either do not respond to Lu177 PRRT or have become resistant to conventional PRRT
So is Targetd Alpha PRRT Available for Patients?
Currently the Nuclear Medicine Therapy Unit at Fortis Memorial Research Institute, Gurugram is the only centre offering targeted Alpha PRRT in India. Targeted Alpha PRRT is available to patients as a salvage therapy with due consent under the Paragraph 37 of the Helsinki Declaration of Therapies of unproven efficacy. What this means is that though there are small studies which prove the efficacy of targeted Alpha PRRT, the quantum of studies is still too small to prescribe Targeted Alpha PRRT as a standard conventional therapy. Nevertheless in patients who have exhausted or are ineligible for conventional therapies and the treating physician as well as the patient is convinced that the patient may benefit from this therapy, Alpha PRRT is offered as an option. The patient’s clinical condition, prior treatments and responses as well as the current clinical situation are meticulously scrutinised by a multidisciplinary tumour board before considering a patient for alpha PRRT. Alpha PRRT is not offered as a clinical trial.
How is it Done?
Just like conventional PRRT, Targeted Alpha PRRT is administered as an intravenous infusion. The patient is admitted in the hospital in a special high dose therapy unit. An intravenous cannula is placed in the forearm. A special amino acid infusion is administered prior to the Alpha PRRT infusion to protect the kidneys. An anti emetic injection is administered as some patients experience nausea due to the amino acid infusion. The alpha PRRT drug is administered over a period of 20 mins.
What are the Adverse Effects?
Targeted Alpha PRRT is very well tolerated. Some patients experience some nausea during the infusion and for a few days after the treatment. Usually the nausea is mild an controlled by taking standard anti nausea medicine. There are some reports of delayed mild derangement of renal function though there are no reports of any patient going into permanent renal failure. There is also practically no haematological toxicity noted.
What are the benefits from Targeted Alpha PSMA Therapy?
After completing almost 50 treatment cycles at the center, the Nuclear Oncology Experts Team is extremely excited with the initial results. With each therapy cycle, most of the patients who have undertaken the treatment have experienced a remarkable improvement in their quality of life with reduced symptoms and pain. Apart from this, the team has also seen an associated biochemical response with a fall in serum PSA levels, indicating a reduction in total tumour burden in the body. Most patients also show a prolonged duration of response with a few patients being in near-complete biochemical and clinical remission for over a year. They continue in clinical remission and as more time goes by the impact of Ac225 PSMA Therapy on the overall survival of patients would become clearer.
The team has performed Ga68 PSMA scans before the next due cycles to monitor radiological responses and have witnessed some encouraging results – nearly 70% tumour reduction in a single cycle in some patients.
Before PSMA Scan with disease in Prostate Gland
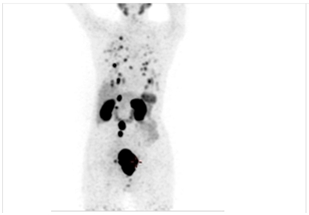
Pre-therapy PSMA Scan with disease in Prostate Gland, Retroperitoneal Lymph Nodes & Lungs (seen as black spots)
After one cycle of Ac225 PSMA Therapy

After one cycle of Ac225 PSMA Therapy, the patient had a remarkable reduction in the active tumour from the lungs (black spots in the image on the left), Retroperitoneal Lymph Nodes, as well as shrinkage of tumour in the Prostate Gland.
What are the side-effects of Targeted Alpha PSAM Therapy?
The most common side-effect of Targeted Alpha PSMA Therapy is dry mouth (Xerostomia), While some patients may find it distressing, most patients do not report a very significant impact on their lifestyle due to the xerostomia.
There is usually no bone marrow suppression, nor any damage to the kidney or liver functions due to Targeted Alpha PSMA therapy.
How are the clinical Outcomes of Alpha PRRT?
Alpha PRRT is administered with a palliative intent. The aim of the treatments is to achieve disease stabilisation with an attempt to improve the progression free survival. In our experience with alpha PRRT over the past two years nearly 75 – 80% patients demonstrate disease stabilisation with most patients reporting a significant improvement in the quality of their lives. There is alleviation of pain, diarrhea with an improvement in the general well being of most patients. Studies have shown durable response with responses continuing for over 30 months after therapy.

Dr. Ishita B. Sen

Fortis Hospital
.
